Transition metal-free photodecarboxylative amination of redox-active esters with diazirines | Organic Chemistry | ChemRxiv | Cambridge Open Engage

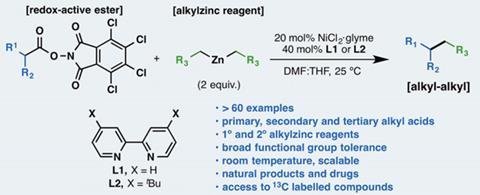
A general alkyl-alkyl cross-coupling enabled by redox-active esters and alkylzinc reagents | Science

N‐(Acyloxy)phthalimides as Redox‐Active Esters in Cross‐Coupling Reactions - Murarka - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library

N‐(Acyloxy)phthalimides as Redox‐Active Esters in Cross‐Coupling Reactions - Murarka - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library

A general alkyl-alkyl cross-coupling enabled by redox-active esters and alkylzinc reagents. - Abstract - Europe PMC
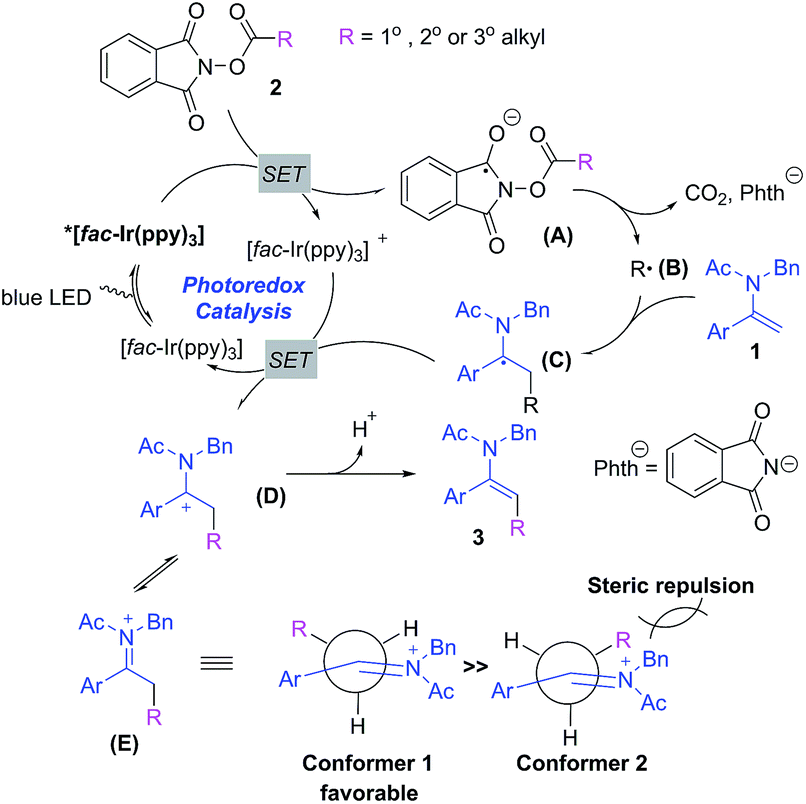
Photoredox-catalyzed stereoselective alkylation of enamides with N -hydroxyphthalimide esters via decarboxylative cross-coupling reactions - Chemical Science (RSC Publishing) DOI:10.1039/C9SC03070K

N‐(Acyloxy)phthalimides as Redox‐Active Esters in Cross‐Coupling Reactions - Murarka - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library

Reactions of copper amine complexes with a redox-active ester Variation... | Download Scientific Diagram

N‐(Acyloxy)phthalimides as Redox‐Active Esters in Cross‐Coupling Reactions - Murarka - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library
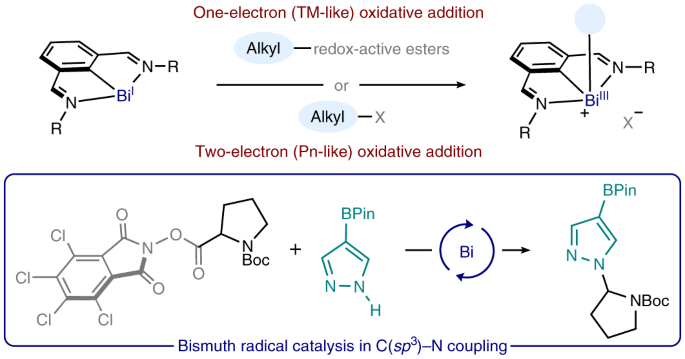
Bismuth radical catalysis in the activation and coupling of redox-active electrophiles | Nature Chemistry
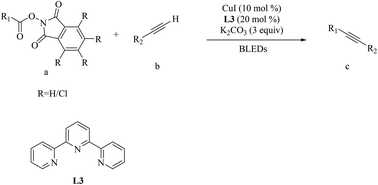
Cu-Photoredox-catalyzed C(sp)–C(sp3) coupling of redox-active esters with terminal alkynes - Organic & Biomolecular Chemistry (RSC Publishing)
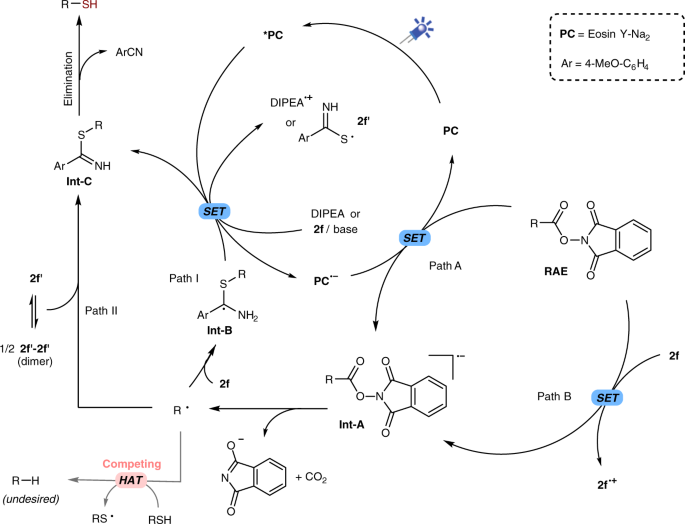
Decarboxylative thiolation of redox-active esters to free thiols and further diversification | Nature Communications

Nickel-Catalyzed Reductive Arylation of Redox Active Esters for the Synthesis of α-Aryl Nitriles – Role of a Chlorosilane Additive | Catalysis | ChemRxiv | Cambridge Open Engage
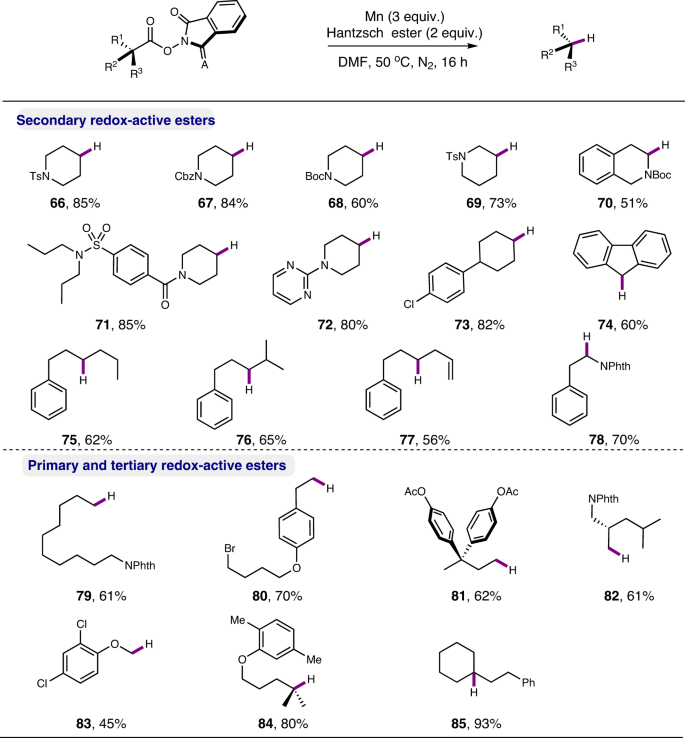
Manganese-mediated reductive functionalization of activated aliphatic acids and primary amines | Nature Communications

Utilization of C(sp3)‐Carboxylic Acids and Their Redox‐Active Esters in Decarboxylative Carbon−Carbon Bond Formation - ScienceDirect